|
|
|
Site Index:
|
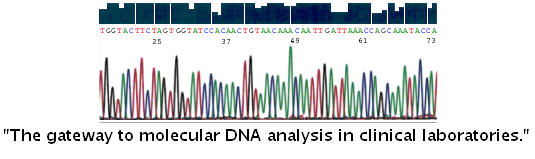
LoTemp™ PCR* with HiFi® DNA polymerase unloading the power of automated Sanger sequencing in DNA testing for Pap cytology specimens Perform DNA sequencing for HPV genotyping beyond a reasonable doubt - BMC Clin Path 2009, 9:3 (22 May 2009). No-false positive Lyme disease DNA testing; Validate the DNA of Neisseria gonorrhoeae opa genes beyond a reasonable doubt; Validate the DNA of Chlamydia trachomatis cryptic plasmid beyond a reasonable doubt; Using the GenBank database as the standard for validating sequence algorithms; On 5% of a liquid-based Pap cytology specimen without need of DNA extraction. These are cutting-edge molecular tests developed by pathologists/ cytotechnologists for pathologists/cytotechnologists to serve gynecologists and their patients at sustainable cost. Application of LoTemp® PCR/DNA sequencing recent publications:
|
|
HiFiDNA.com © 2006-2013 |